Consider VPRIV
VPRIV is indicated for long-term enzyme replacement therapy (ERT) for patients with type 1 Gaucher disease and has established safety and efficacy data in patients aged 4 years and older, who were in various stages of life. Whether you’re considering treatment for yourself or for a loved one, choosing the right one is a vital part of any patient’s experience with type 1 Gaucher disease. To help understand if VPRIV is the right choice for you or a loved one, consider the following:

VPRIV has over 10 years of real-world experience. VPRIV was first approved by the FDA in 2010 and has been indicated for long-term use to treat patients with type 1 Gaucher disease ever since.

VPRIV was studied in the largest clinical trial program of an ERT for type 1 Gaucher disease; up to 99 patients in various life stages (4 years and older) were evaluated across three clinical trials and a long-term extension study. Learn more about these studies here.

VPRIV is an enzyme replacement therapy, specifically designed to match and replace the natural human enzyme (glucocerebrosidase) that is missing with type 1 Gaucher disease. VPRIV is an ERT for GD1 that is made from a human cell line; this design is intended to facilitate targeted uptake of VPRIV into cells.

For patients 4 years and older who are new to treatment, VPRIV is administered as a 60-minute infusion, taken once every other week under the supervision of a healthcare professional.
If you and your doctor determine that VPRIV is right for you, there are support programs to help you during your journey. Click here to learn more.
What Is VPRIV?
VPRIV is an ERT (enzyme replacement therapy) indicated for long-term use for patients with type 1 Gaucher disease. VPRIV was evaluated in pediatric, young adult, and older adult patients aged 4 years and older in the largest ERT clinical trial program for GD1, where safety and efficacy were established.
People with type 1 Gaucher disease are missing, or have low activity of, the enzyme glucocerebrosidase (GLOO-ko-SER-e-bro-si-daze). VPRIV is designed to help the body address this deficiency and is the only ERT for GD1 that is made from a human cell line, meaning it has the same protein structure as the naturally occurring enzyme. This feature is designed to facilitate targeted uptake of VPRIV into cells.
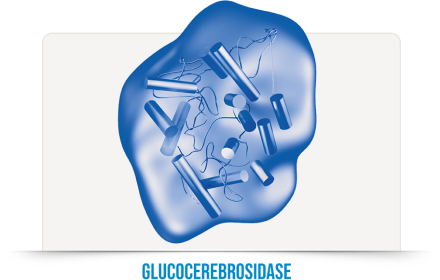

As patients with GD1 are missing the enzyme glucocerebrosidase, VPRIV is specifically designed to match and replace the natural human enzyme, replicating its job: to remove excess glucocerebroside (GLOO-ko-SER-e-bro-side), a fatty substance that builds up in cells and causes them to enlarge.
VPRIV works by binding to and being absorbed into cells affected by type 1 Gaucher disease. Once inside the cell, VPRIV breaks down the glucocerebroside to reduce the overall amount.
Learn more about the clinical trials for VPRIV treatment here.
How Does VPRIV Work?
1
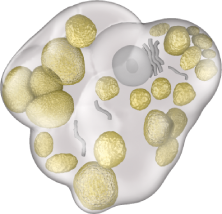
Gaucher cells are swollen due to the accumulation of a fatty substance known as glucocerebroside.
GAUCHER CELL
2
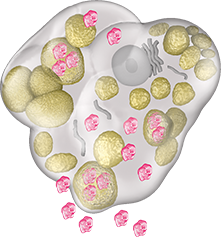
VPRIV is absorbed into the
cell. Once inside the cell,
VPRIV breaks down
glucocerebroside.
Vpriv action
3
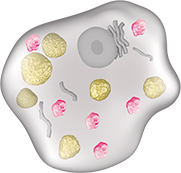
Like the naturally occurring human enzyme, VPRIV breaks down glucocerebroside, reducing the overall amount in the cell.
resulting cell
For illustration purposes only. In vitro test results do not necessarily correlate with clinical efficacy.
Ask your doctor if VPRIV could be part of your GD1 treatment plan.

